Martínez-Romero GJ1, Nagore Gallego MD2, Gómez Marín R3, Serrano Olaizola A4
1 Pharmacy Department. Manises Hospital. Valencia (Spain)
2 Geriatric Unit. Department of Internal Medicine. Manises Hospital. Valencia (Spain)
3 Pharmacy Department. Quirón Torrevieja Hospital. Alicante (Spain)
4 Orthopedic Surgery Department. Manises Hospital. Valencia (Spain)
Fecha de recepción: 23/04/2019 – Fecha de aceptación: 02/06/2019
Correspondencia: Hospital de Manises (Servicio de Farmacia) – Avda. de la Generalitat Valenciana, 50 – 46940 Manises. Valencia (España)
gmartinez@hospitalmanises.es
____
SUMMARY
Key Words: Teicoplanin, pharmacokinetic monitoring, prosthetic infection.
We report a patient with chronic knee prosthetic infection, underwent multiple surgical procedures and previous antibiotic treatments. Traumatologist indicated chronic suppressive antibiotic treatment and no further surgery was indicated due to the patient´s high comorbidity. A microbiology examination revealed the presence of hetero-resistant Staphylococcus epidermidis strains, which meant the possibility of being treated only with linezolid or glycopeptides. It was discarded a chronic use of linezolid due to reasons of hematological toxicity and optical and peripheral neuropathy described in treatments over 28 days. Teicoplanin was proposed for good pharmacokinetic profile, which makes it optimal for outpatient management regimes. Monitoring of treatment was based on the pharmacokinetics monitoring teicoplanin trough levels, and dose adjustment based on them. Monitoring inflammation parameters, hematology and renal function was performed to monitor the effectiveness and toxicity of treatment. During the follow-up period (18 months) patient presented a satisfactory clinical evolution without notable toxicity. In this sense, in selected patients, teicoplanin with outpatient management may be useful in treating chronic infections of joint prosthesis.
Monitorización farmacocinética ambulatoria de teicoplanina por infección crónica de prótesis de rodilla
RESUMEN
Presentamos el caso de un paciente con infección crónica de prótesis de rodilla, sometido a múltiples intervenciones quirúrgicas y tratamientos antibióticos previos. La indicación de Traumatología es tratamiento antibiótico de supresión crónico, contraindicando nueva intervención quirúrgica. En esta situación, se constata que la infección está causada por una mezcla de cepas de Staphylococus epidermidis con heterorresistencia con sensibilidad únicamente a linezolid y a glupopéptidos. Se descartó el uso crónico de linezolid por razones de toxicidad hematológica y neuropatía óptica y periférica descritos en tratamientos superiores a 28 días. Se propuso teicoplanina por su buen perfil farmacocinético, que la hace óptima para regímenes de administración ambulatoria. El seguimiento del tratamiento se basó en la monitorización farmacocinética de los niveles valle de teicoplanina, y ajuste de dosis en función de los mismos. Se realizó seguimiento de parámetros de inflamación, hematológicos y de función renal para monitorizar la efectividad y toxicidad del tratamiento. El paciente presentó durante el periodo de seguimiento (18 meses) una evolución clínica satisfactoria sin toxicidad destacable. En este sentido, en pacientes seleccionados, teicoplanina administrada por unidades ambulatorias de administración de antibióticos puede resultar útil en el tratamiento de infecciones crónicas de prótesis articulares.
Palabras clave: Teicoplanina, monitorización, niveles valle, infeccion prótesis articular.
____
INTRODUCTION
Teicoplanin is an antibacterial glycopeptide against gram+ cocci whose clinical efficiency correlates with permanence over the MIC, especially with the AUC24h/MIC value. For this reason, it is important to monitor trough concentration and individually adjust doses to keep trough levels above 10 µg/mL. Teicoplanin offers the ideal pharmacokinetic characteristics (t1/2: 100-170h) to be used by hospital home care units (HHCU) in an ambulatory setting.
We present a case of an 83-year-old man with high blood pressure, dyslipidaemia, COPD, hypertensive cardiopathy, brady-tachycardia syndrome with paroxysmal atrial fibrillation, and heart failure and secondary respiratory episodes, chronic kidney failure and hypothyroidism.
In June 2014, he came to our hospital about his left knee infection, operated on 10 occasions. It was verified that he had arthrodesia infection, and that two fistulae were present for which treatment with cotrimoxazole was administered. Amputation was considered, but the patient preferred chronic antibiotic treatment and fistula cleansing. He remained stable until July 2016, when the fistulae became worse and withdrawing the osteosynthesis material by surgical cleaning was considered. He underwent surgery in February 2017 and a stud from the arthrodesia was removed, while a spacer with vancomycin and gentamicin was placed. While still in hospital, he started treatment with rifampicin and vancomycin, plasma concentration was monitored and the individual dose was adjusted.
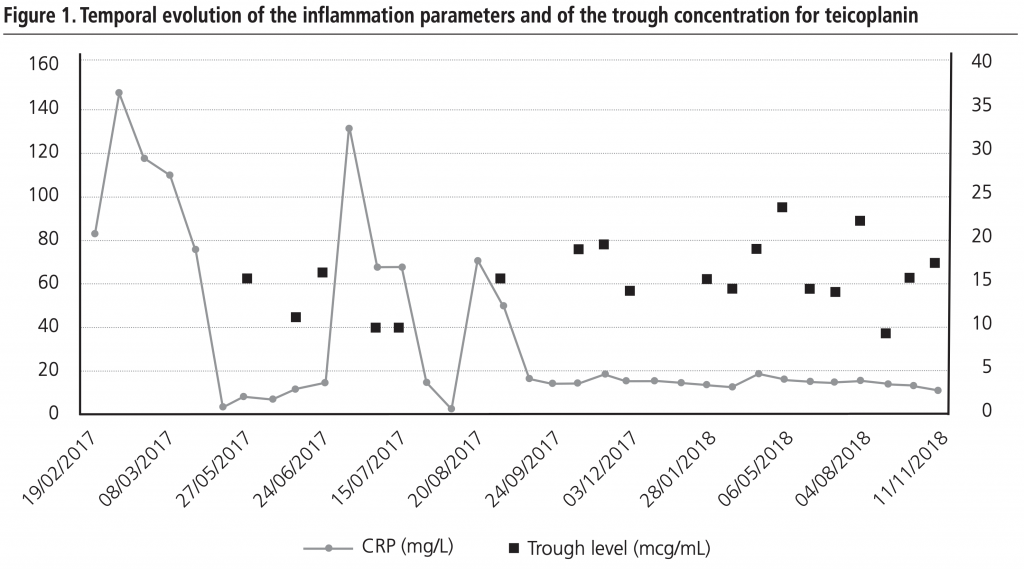
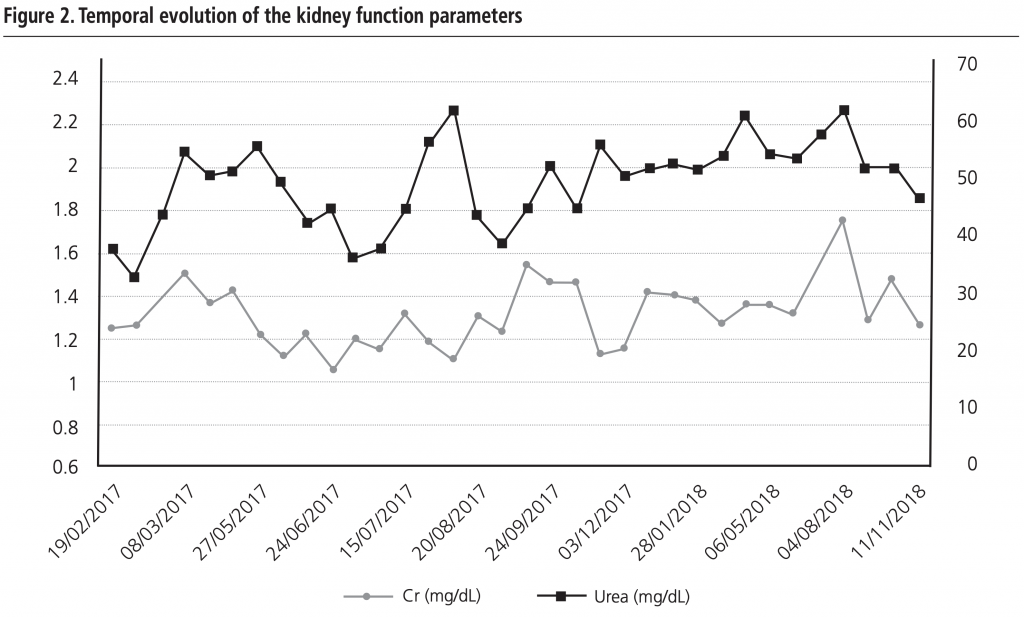
A microbiology examination revealed the presence of hetero-resistant Staphylococcus epidermidis strains, which meant the possibility of being treated only with linezolid or glycopeptides. Traumatologist indicated chronic suppressive antibiotic treatment given the risk of infection persisting, and no further surgery was indicated due to the patient’s high comorbidity. The patient was placed on oral linezolid treatment, 600 mg/12h, but because of his chronic treatment indication, other options were assessed by the multidisciplinary team involved (geriatrician, traumatologist, a doctor from HHCU and a pharmacist) since haematological1 and neuropathic2 toxicity, along with high cost3, (although now, with the generic presentation this is not a problem) would limit its chronic use. After being on linezolid treatment for two and a half months, treatment was changed to teicoplanin, administered through the HHCU at the patient’s home, along with pharmacokinetic monitoring in the Pharmacy Service. The guidelines of the BSAC European OPAT Summit Conference of March 2011, by the Greater Glasgow and Clyde NHS, were followed. This consisted in administering a loading dose according to creatinine clearance and the patient’s weight, followed by a dispensing plan of 3 days/week (Mondays, Wednesdays and Fridays) with dose based on kidney function. After the initial phase, trough levels were first determined, which fell within the therapeutic range (trough level: 15.5 µg/mL). This pattern was later altered to two days/week (Mondays and Thursdays) and the trough concentration (trough level) after 3 weeks was 10.7 µg/mL. At this point of the treatment, a decision was made to ensure viable treatment and one that the patient would accept, which involved attempting weekly administering teicoplanin. A search in the literature was done and one similar clinical case4 was found. A decision was made to weekly administer 2,800 mg of teicoplanin, and to monitor trough level fortnightly and kidney function weekly, plus doing a blood count and monitoring inflammation parameters (CRP/ESR). During the 18-month follow-up, the mean trough level value for teicoplanin was 16.03 µg/mL (within the range of 9.2-24.6 µg/mL; Figure 1). At the beginning, doses were adjusted according to trough level and kidney function to target 15 µg/mL. Then we incorporated the population pharmacokinetic model published by Ogawa et al.5 and through the NONMEM programme, v.7.3.0 (non-linear mixed effects model). Individual PK parameters were estimated with the Bayesian approach, with which different dose adjustment scenarios were simulated to help with decision making. The patient’s evolution has been satisfactory as no sign of inflammation has appeared and the fistulae cured. Two hospitalisations for decompensation of the patient’s heart pathology with high associated CRP/ESR (Figure 1) are noteworthy, but his knee was inflammation-free. Figure 2 illustrates kidney function evolution in creatinine and serum urea terms. His worsened condition in summer was due to dehydration from not drinking enough water. This was corrected, baseline levels were restored, and no significant worsening was evidenced. Haematologically speaking, the values for the following remained stable; haemoglobin (mean:12.7 g/dL range: 11.8-13.6 g/dL), leukocytes (mean: 5.7×103 range: 4.4-7.2×103) and platelets (mean:180×103 range:122-271×103); and no toxicity was seen. The patient is still (November 2018) on the weekly intravenous teicoplanin treatment, adjusted according to individual PK parameters, estimated to obtain a trough level of 10-15 µg/mL. Based on this clinical case and the aforementioned one, 4 teicoplanin, along with pharmacokinetic and toxicity monitoring, is an alternative in patients with chronic osteoarticular infection by Gram+ in whom treatment with oral antibiotics for toxicity and/or resistances is not feasible.
Conflict of interests: The authors declare no conflict of interests.
BIBLIOGRAPHY
1. Cattaneo D, Orlando G, Cozzi V, Cordier L, Baldelli S, Merli S, Fucile S, Gulisano C, Rizzardini G, Clementi E. Linezolid plasma concentrations and occurrence of drug-related haematological toxicity in patients with gram-positive infections. Int J Antimicrob Agents. 2013;41:586-9.
2. Bressler AM, Zimmer SM, Gilmore JL, Somani J. Peripheral neuropathy associated with prolonged use of linezolid. Lancet Infect Dis. 2004;4:528-31.
3. Dilip Nathwanil, Gavin D. Barlow, Katherin Ajdukiewicz, et al. Cost-minimization analysis and audit of antibiotic management of bone and joint infections with ambulatory teicoplanin, in-patient care or outpatient oral linezolid therapy. Journal of Antimicrobial Chemotherapy. 2003;51:391-396.
4. Long-term, once-weekly outpatient teicoplanin use for suppression of chronic prosthetic joint infection. Letters to the editor. Int J. Antimicrob Agents. 2013; 41:197-202.
5. Ryuichi Ogawa, Seiichi Kobayashi, Yuki Sasaki, Mizue Makimura, Hirotoshi Echizen. Population pharmacokinetic and pharmacodynamic analyses of teicoplanin in Japanese patients with systemic MRSA infection. Int J Clin Pharm TH. 2013;5:357-66.
____
